Organizations use document control software systems to manage and automate critical documentation in a more secure and efficient way. Companies in life sciences – including biotech, healthcare, pharmaceutical, and more – use it to simplify many burdensome processes while staying compliant.
Read on to discover how organizations like yours are using document control software to save time and effort – and, therefore, costs.
What Is Document Control?
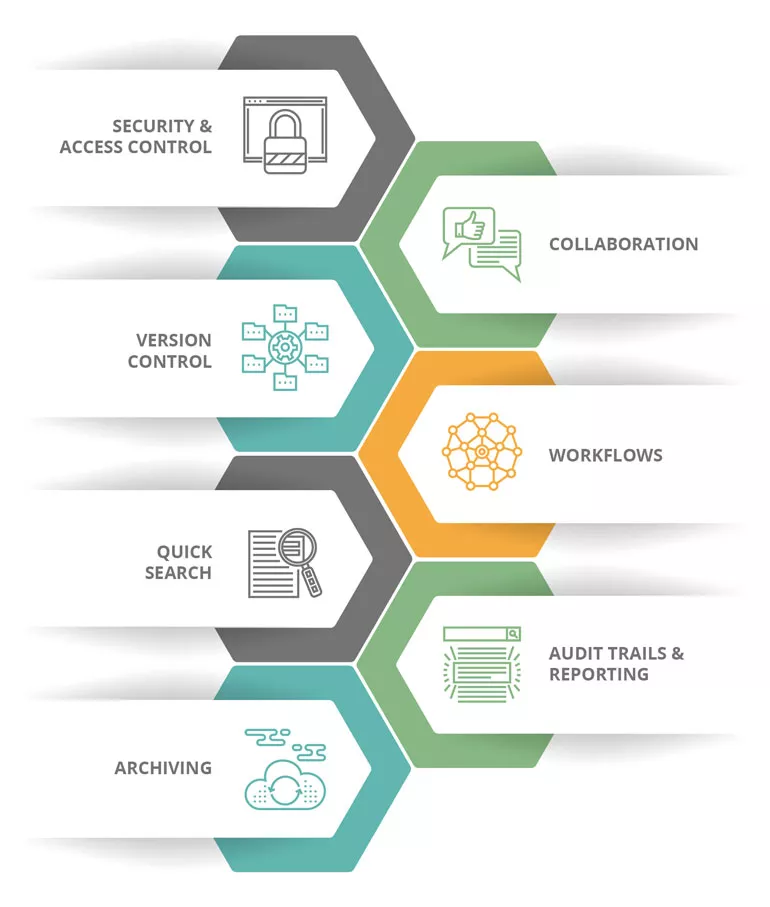
Document control is the process by which organizations manage documents from the point of creation to distribution and storage. These systems encompass workflows that include your documents themselves, the protocols you’ve put in place to control them, and who gets to interact with them.
Why Do Companies Need Document Management System Software?
Organizations need document control software to ensure compliance, security, productivity, and efficiency. A document control system centralizes a company’s documentation in a single software-based platform, automating the management of all files & documents and their workflows. This centralization and automation enable organizations to spend less time searching for and processing documents, keeping document-related workflows adherent to industry regulations.
How to Improve Efficiency and Compliance With Document Control?
The manual ways of digitally managing documents simply don’t cut it in today’s digital environment. Whether you use document control software on your premises or in the cloud, you’ll see the following benefits:
Maintain Regulatory Compliance
Document control software systems allow you to automate your standard operating procedures (SOPs) through workflows and demonstrate they’re being followed. An electronic training module allows you to automatically assign content/compliance-based training by job title and produce training reports for auditors with the touch of a button. The technology enables you to prove that your sensitive documents are controlled properly and accessed by only the right people. When audit time comes around, the digital storage of your documents will help you retrieve the relevant ones instantly and organize the material needed to quickly collect data for the audit.
Increase Efficiency and Reduce Overhead
Using a document control software system enables your organization to lower overhead costs through several benefits. Your business will save time by not dealing with manual, paper-based processes. Employees who are free from having to chase documents through workflows will get their day back and deliver efficiency through work instead of stagnating productivity. You’ll also see the cost-saving benefits of sustainability – with paper out of the picture, you’ll save on ink and all products related to paper processes.
Improve Quality Management Processes
Document control software ties together your quality processes – making them traceable and reportable so that decision-makers can identify both efficiencies and areas for improvement. Additionally, your SOPs, policies, and other quality processes better adhere to regulations while you manage change control and incidents.
How Document Control Software Works
DocXellent’s document control software, ENSUR, is built around open technologies that allow you to integrate with your existing applications and maximize your current systems’ potential. ENSUR document management is accessible by your global workforce – no matter where they are or what device they use.
Here’s what you can do with ENSUR:
Automate Manual Document Workflows
The document control software streamlines review, approval, distribution, retrieval, and obsolescence processes for documents. You’ll gain revision control and revision history tracking while being able to enforce document standards. You’ll also be able to automate the periodic review of aging documents.
Improve Audit Readiness
Enjoy easily searching for and finding documents come audit time. The software provides comprehensive audit trails and reports, and you can quickly produce employee training records. If your industry requires FDA 21 CFR Part 11 compliant e-signatures, tthe document controlling software also captures those and manages your entire audit process, including audit response, assigning action items, and producing reports.
Centralize and Improve Collaboration
ENSUR’s DMS enables controlled access to documents in a global workforce, including employees, suppliers, and contractors. People who have secure access to documents are always supplied with the most recent version – eliminating multiple communication channels and potentially wasted time with older documents.
Maintain Complex Supply Chains and Mitigate Risks Therein
Document control software systems give you instant visibility into documents located in supply chain-related workflows. You’ll be able to manage specifications more easily and remove entry points of error in those workflows.
Business Continuity and Disaster Recovery
ENSUR’s cloud-based document management system allows for seamless uptime in the event of a disaster, and you keep peace of mind knowing your critical business data remains secure. There is no risk of physically losing documents, and your employees maintain access to files should a cyberattack or natural disaster occur.
Testimonials
“The system is easy to implement and use because we have excellent support from DocXellent! They have always been great to work with and make hard tasks easy.” Richard V. Change Control Analyst Mesa Labs
“One of the best customer support teams we’ve worked with. Outstanding product and smart updates.” Chris M. Senior Director Denison Pharmaceuticals
“We implemented ENSUR in December 2019. Being fully remote for two years, starting in March 2020, we rely on ENSUR for more and more all the time. We could not have handled our research and training documentation during the pandemic without ENSUR and the DocXellent team. We have used ENSUR for a vendor audit and were able to provide all requested documentation with ease.” Melissa M. Intranet and Quality Management Software Administrator Harvard Research
“ENSUR has dramatically reduced the number of errors and miscommunications with controlled documents and forms. If we ever have an issue, question, or just a suggestion, ENSUR customer service is always listening and eager to assist. To date, it remains one of our most successful system launches impacting almost every employee.” Albert Webster Computer Software Validation Coordinator Ortho Molecular Products
“DocXellent’s Support team is fantastic – super responsive, pleasant to deal with, helpful, and informative; they often try to offer more than one way to solve a problem. DocXellent’s monthly newsletter and frequent webinars provide useful insights into features of ENSUR that we may not currently be using.” Alexandra C. Quality Systems Manager International Vitamin Corporation
“Utilizing ENSUR has helped us achieve a database for all our documentation, training records, and processes. Having all of these in one location helps us achieve an audit trail and historical records for future reference. I also love the support given. The DocXellent team are truly some of the most caring, helpful, and personable people I know. They are always a click away and ready to help.” Jamie A. Quality Assurance Specialist Nutrabolt
“ENSUR has enabled us to streamline our document change management process. We now have version control and are able to control the review and approval process, track user engagement, and produce reports for our auditors. The best part of working with ENSUR is the team at DocXellent. From the engineers to the trainers, everyone is so helpful and responsive. I would definitely recommend ENSUR to anyone considering document control software.” Kristen A. Executive Assistant to CEO Colonial Management Group LP
Case Studies
CASE STUDY-MESA LABS v1.0 CASE STUDY-ORTHOMOLECULAR v1.0 CASE STUDY-IVC v1.0 CASE-STUDY-PHARMA-CMO v1.0 CASE STUDY-AMGRAPH v1.0 Grant Industries Increases Revenue and Streamlines Audits With ENSUR
Frequently Asked Questions
- What is Document Control?
- Document Control is the practice of enforcing standardized, controlled processes for managing your documents from the point of creation to distribution and storage.
- What is the document control process?
- The document control process is the workflow of your documents as defined by your company’s policies, including types of documents involved, document identification and versioning, document authoring, controlled and uncontrolled document copies, review and approval routing, storing and retrieving, access control and distribution, retention and disposal, and change control.
- What are document controllers responsible for?
- A document controller is responsible for managing and executing a company’s document control process, ensuring that all documents comply with company policies and standards.
- Why is it important to have document control?
- Document control is a critical piece of your company’s quality assurance system because it ensures your documentation is accurate, organized, traceable, reportable, and compliant with any legal or regulatory standards.
- What are document control procedures?
- Document control procedures are the series of actions your company has established for managing your documents throughout their workflow.
- What is the difference between document control and document management?
- Document control refers to the way documents flow through your organization. Elements of document control include approvals, revisions, and version control. As documents move through workflows in a business, they need to be controlled within that flow of knowledge — that’s what document control is for. Document management refers to storing and distributing documents, including retrieval, tracking, and sharing.
- What are the most regulated industries in terms of document control?
- While many industries have their fair share of regulations to abide by, the medical device manufacturing, life sciences, and pharmaceutical industries are three of the most highly regulated when it comes to document control.
- What are the key benefits of document control software?
- Document control software delivers a multitude of benefits to small and large organizations in any industry:
- With a centralized platform to manage documents, employees can do more strategic work as they won’t have to spend time sifting through paper files or locally-stored drives.
- With document file management, you are rest assured of greater document security with tools such as access control and encryption.
- Version tracking and audit trails ensure your compliance and make the process of compiling documents much faster in case of an audit.
- You cut costs on all physical equipment and space needed to store and process documents, including filing cabinets, copiers, ink, folders, and more.
- Your team sees a boost in productivity and efficiency as searching for, retrieving, and storing documents can be done instantly in document control software.
- Document control software delivers a multitude of benefits to small and large organizations in any industry:
If you’re ready to simplify your company’s paper document processes in a more secure and efficient way, you’ve come to the right place. With digital document control software from DocXellent, you can improve collaboration, workflows, and more. Request a demo today and see how DocXellent can transform your most burdensome daily tasks!




























