Many companies are still using spreadsheets, shared drives or outdated systems to manage the packaging lifecycle. These manual systems put your processes at high risk for human error and inefficiencies causing time and money loss and delays to market.
We hear the same common pain points from packaging professionals across the globe:
- Too easy for packaging engineers to use the wrong version of a spec
- Hard to manage “common specs” with uncontrolled values cause inconsistencies across specs
- Using email to get approvals is error-prone and causes time delays
- Outdated versions of specs are being sent to suppliers
- Inefficient processes for linking related specs, technical drawings, die lines, and reference documents
- Precious time is spent trying to find the impact of a spec change with regards to the BOMs it is associated with
- Searching across fields for common traits is difficult and time-consuming
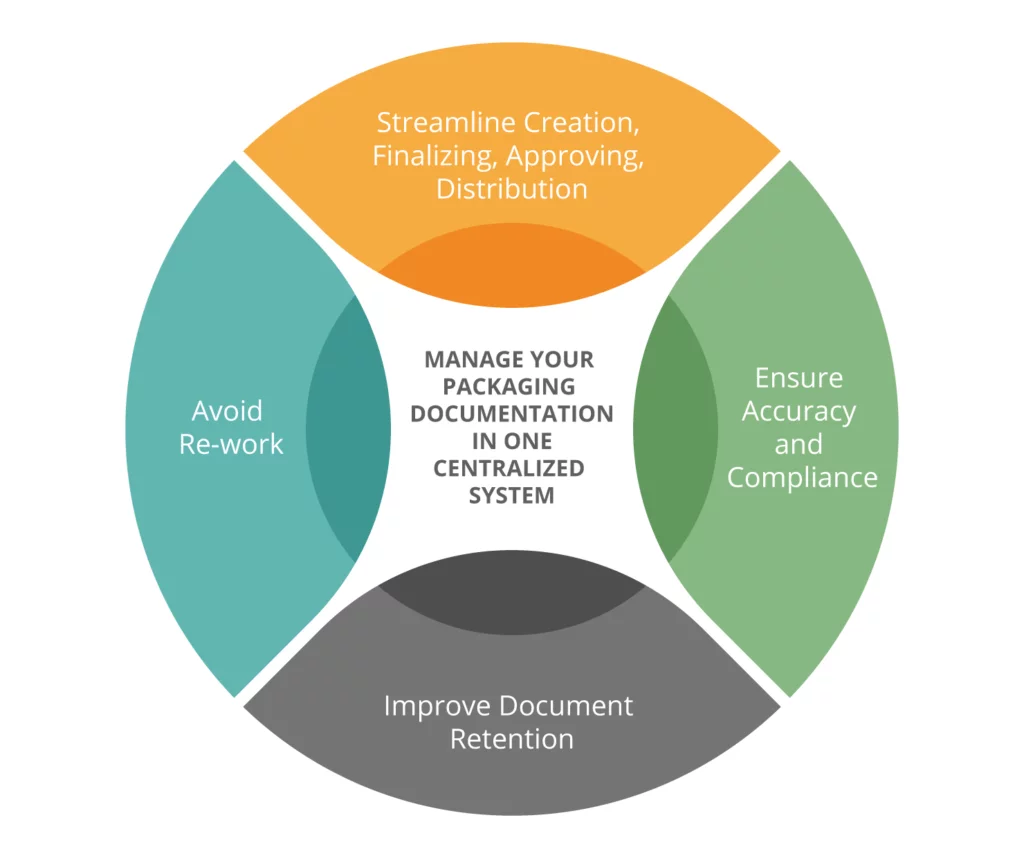
ENSUR – One Platform for Managing All of Your Packaging Documentation
The ENSUR Specification Management Software streamlines the creation, finalizing, approval and distribution of packaging specifications and documents into one platform, providing game-changing collaboration and automation capabilities. Using one centralized system for managing your packaging specs and documentation, you can easily align your different departments on standards to ensure quality and compliance, significantly increase process efficiency, and reduce time to market.
ENSUR is a user-friendly, browser-based system that is available on-premises or in our GxP compliant Cloud so that your employees and suppliers/CMOs can access the system anytime, anywhere. Fully integrated with our document management features, the document management software allows for complete control over who can access which documents and tracks all activities with the ability to create reports for visibility and compliance. ENSUR’s version control features and flexible security model guarantee that your employees and suppliers are always working from the correct version of a spec or document. The system integrates with your email client to send email notifications to internal and external stakeholders at each step of the workflow, improving collaboration and removing bottlenecks from your processes.
ENSUR Gives You the Power to:
Search & Find: Instantly find common traits among your products and locate where components are used in your product line
Add Identifying Attributes: Add critical attributes to specs for identifying customers, locations and quantities
Notify: Automated email notifications to suppliers and stakeholders when specs and docs become effective
Report: Produce reports with attachments and subordinate specs/references/drawings and configure reports for different audiences
Control Versioning of Specs: Create, revise and replicate specs in a controlled environment that prevents human error and ensures the correct version of a spec is being used and produced
Create BOMs & Finished Goods: Easily create BOMs and Finished Goods and manage both component qty/units AND packaging information (palletization, master data, etc) all in one specification
Collaborate with Suppliers: Collaborate with suppliers and CMOs from anywhere in the world on specs in a controlled environment and manage which suppliers have access to specific documents
Migrate Your Data from Legacy Systems
DocXellent makes it easy to migrate data from your legacy systems into newer, more flexible out of the box technology. Regardless of the record, (including Change Requests, Packaging Specifications, Quality Assurance Procedures, Product Specifications and BOMs) it can be migrated into ENSUR. Our technical team has developed proven methods to migrate your data into ENSUR without data loss or compromise, so you can continue to use your business content with confidence going forward. This approach optimizes the rollout period and gets your team using your legacy data and documents in the new system sooner than expected.
- What is Pack Spec Management?
- Pack Spec Management is the process of managing your packaging specifications within a centralized system where you can 1) control and approve the values and inputs of your specs through structured data and 2) automate the review, approval and distribution processes of specs to stakeholders.
- What is a packaging specifications software system?
- A packaging specification software system is a comprehensive platform for creating, maintaining and publishing your packaging specifications and related documents. These systems also utilize document control features, which give you the ability to create reports for compliance and allow you to have complete control over who can access which documents.
- What are the 4 main components of packaging specification management software?
- The four main components of packaging specification management software are:
- Specifications and Bill of Materials (BOM) Management
- Vendor/Supplier Management
- Searching and Reporting
- Integration with ERP
- The four main components of packaging specification management software are:
- What is version control?
- Version control is a process that keeps track of every modification made to a specification or document within your pack spec system. Version control helps teams mitigate issues that may arise from working from different versions of a specification, tracking every individual change by each contributor and helping prevent concurrent work from conflicting.



































